From Dr. McMunn’s Elixir of Opium, sold in the 1830s, to the state of Colorado legalizing recreational marijuana, the U.S. has a long history of government drug regulation. Current federal law defines 5 separate categories or “schedules” for drugs. Substances are classified by their acceptable medical use and potential for dependency and abuse, though never without controversy.

Addictive drugs were first outlawed in the U.S. after the International Opium Convention, held in January 1912. Concerned about a thriving global opium trade, 13 nations resolved to “…use their best endeavors to control or to cause to be controlled all persons manufacturing, importing, selling, distributing, and exporting morphine, cocaine, and their respective salts.”
Not until 1970 with the passage of the Controlled Substances Act, signed into law by then President Richard Nixon, did the present-day system of drug schedules go into effect. The phrase “controlled substance” means a drug or other substance, or a derivative thereof, included in one of the 5 schedules.
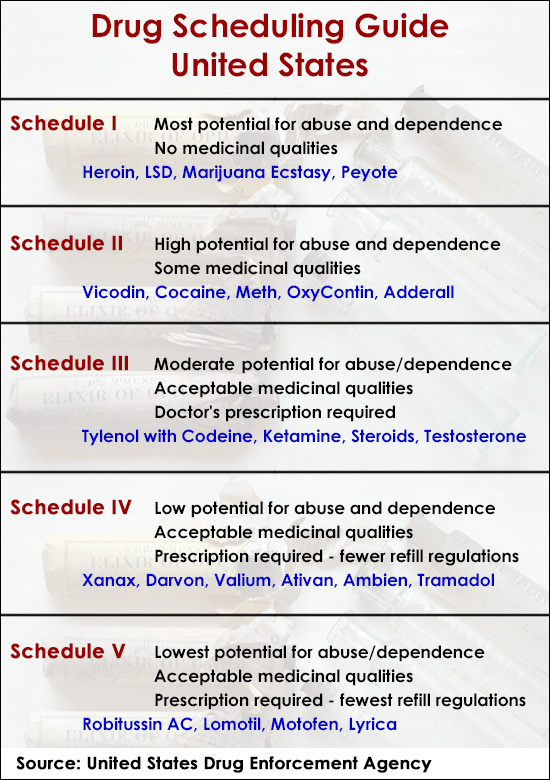
What are the Five Drug Schedules in the United States?
Schedule I Drugs
Schedule I drugs are the most dangerous drugs of all 5 categories. These substances with potentially severe physiological and psychological dependence are considered to have no acceptable medicinal qualities. Additionally, you cannot get a prescription for any drugs falling into this category. Here’s a list of some of the schedule I drugs:
- Heroin
- Marijuana
- MDMA (ecstasy)
- Lysergic acid diethylamide (LSD)
- GHB (date rape drug)
- Mescaline
- Cathinone (used in bath salts)
- Peyote
- Psilocybin and psilocin (mushrooms)
Schedule II Drugs
Schedule II substances are also commonly abused and cause dependency. However, these drugs have a legitimate use in medical treatment, though many of them come with severe restrictions. This class of drugs is only legal when prescribed to an individual by their doctor and received from a pharmacist. Here are some of the schedule II substances:
- Cocaine
- Methamphetamine
- OxyContin
- Adderall
- Ritalin
- Dilaudid
- Methadone
- Fentanyl
- Dexedrine
- Vicodin
Schedule III
Includes those drugs believed to have a moderate to low potential for physical and psychological dependence and have accepted medicinal qualities. Like schedule II, these substances may only be legally obtained with a doctor’s prescription. Some examples of Schedule III drugs are
- Ketamine (considered a date rape drug)
- Anabolic steroids
- Buprenorphine (Suboxone, Subutex)
- Testosterone
- Tylenol with codeine
Schedule IV Drugs
still require a prescription, though physicians are allowed to include refills up to five times in a 6-month period without additional consultation. Additionally, these are substances thought to have a relatively low potential for abuse and addiction. Schedule IV drugs include the following:
- Benzodiazepines (Xanax, Valium, Klonopin)
- Benzodiazepine-like drugs (Ambien, Sonata, Lunesta)
- Phenobarbital (long-acting barbiturate)
- Modafinil (Stimulant-like drug)
- Alprazolam (Xanax)
- Diazepam (Valium)
- Lorazepam (Ativan)
- Clonazepam (Klonopin)
- Tramadol
- Zolpidem (Ambien)
- Eszopiclone (Lunesta)
- Temazepam
Schedule V
Substances have the lowest potential for abuse among all controlled substance categories. Although they still require a prescription, refills may be authorized by the prescribing physician. Some examples of Schedule V drugs include:
- Cough suppressants with codeine (Robitussin AC)
- Pyrovalerone (used for chronic fatigue and appetite suppressant)
- Some anticonvulsants (Lyrica, Vimpat)
Information provided by the Drug Enforcement Administration
Both the Drug Enforcement Administration (DEA) and the Food and Drug Administration (FDA) determine which substances are added or removed from each schedule. These agencies have come under fire in recent years for a myriad of reasons, including taking too long to approve new medications and not removing substances, like marijuana, that states are making legal.
Critics of the current drug schedules have long pointed out the absence of alcohol and tobacco, the most widely used and abused drugs in the United States. Some have argued that this is an important exemption considering the tax revenue derived from these substances.
Related:
Law Enforcement Assisted Diversion (LEAD) Could End War On Drugs
Prescription Drug Monitoring Program
The Dangers of Abusing Ketamine





